COVID-19 vaccination and boosting during pregnancy protects infants for six months
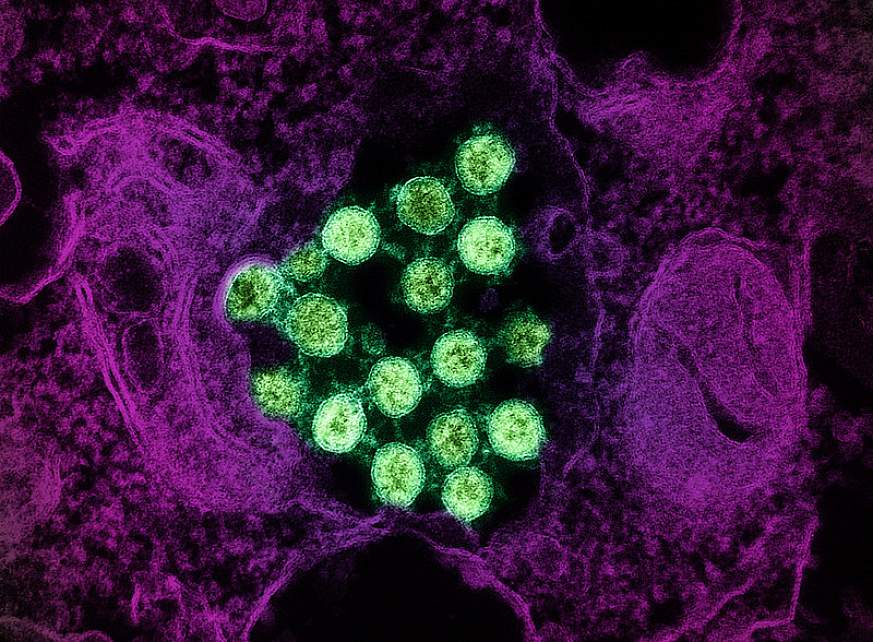
Transmission electron micrograph of SARS-CoV-2 virus particles (colorized green), isolated from a patient sample.NIAID
What
Women who receive an mRNA-based COVID-19 vaccination or booster during pregnancy can provide their infants with strong protection against symptomatic COVID-19 infection for at least six months after birth, according to a study from the National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health. These findings, published in Pediatrics, reinforce the importance of receiving both a COVID-19 vaccine and booster during pregnancy to ensure that infants are born with robust protection that lasts until they are old enough to be vaccinated.
COVID-19 is especially dangerous for newborns and young infants, and even healthy infants are vulnerable to COVID-19 and are at risk for severe disease. No COVID-19 vaccines currently are available for infants under six months old. Earlier results from the Multisite Observational Maternal and Infant COVID-19 Vaccine (MOMIv-Vax) study revealed that when pregnant volunteers received both doses of an mRNA COVID-19 vaccine, antibodies induced by the vaccine could be found in their newborns’ cord blood. This suggested that the infants likely had some protection against COVID-19 when they were still too young to receive a vaccine. However, researchers at the NIAID-funded Infectious Diseases Clinical Research Consortium (IDCRC), which conducted the study, did not know how long these antibody levels would last or how well the infants would actually be protected. The research team hoped to gather this information by following the infants through their first six months of life.
In this portion of the study, researchers analyzed data from 475 infants born while their pregnant mothers were enrolled in the MOMI-Vax study. The study took place at nine sites across the United States. It included 271 infants whose mothers had received two doses of an mRNA COVID-19 vaccine during pregnancy. The remaining 204 infants in the study were born to mothers who had received both doses of an mRNA COVID-19 vaccine as well as a COVID-19 booster. To supplement data gathered during pregnancy and at birth, the infants were evaluated during at least one follow-up visit during their first six months after birth. Parents also reported whether their infants had become infected or had demonstrated COVID-19 symptoms.
Based on blood samples from the infants, the researchers found that newborns with high antibody levels at birth also had greater protection from COVID-19 infection during their first six months. While infants of mothers who received two COVID-19 vaccine doses had a robust antibody response at birth, infants whose mothers had received an additional booster dose during pregnancy had both higher levels of antibodies at birth and greater protection from COVID-19 infection at their follow-up visits.
While older children and adults should continue to follow guidance from the Centers for Disease Control and Prevention (CDC) to stay up-to-date on their COVID-19 vaccines and boosters, this study highlights how much maternal vaccination can benefit newborns too young to take advantage of the vaccine: During the course of this study, none of the infants examined required hospitalization for COVID-19. Researchers will continue to evaluate the data from the MOMI-Vax study for further insights concerning COVID-19 protection in infants.
Article
CV Cardemil et al. Maternal COVID-19 Vaccination and Prevention of Symptomatic Infection in Infants. Pediatrics DOI: 10.1542/peds.2023-064252 (2024).
Who
Cristina Cardemil, M.D., a medical officer in NIAID’s Division of Microbiology and Infectious Diseases, is available to comment.
Contact
To schedule interviews, please contact Elizabeth Deatrick, (301) 402-1663, [email protected].
NIAID conducts and supports research—at NIH, throughout the United States, and worldwide—to study the causes of infectious and immune-mediated diseases, and to develop better means of preventing, diagnosing and treating these illnesses. News releases, fact sheets and other NIAID-related materials are available on the NIAID website.
About the National Institutes of Health (NIH):
NIH, the nation’s medical research agency, includes 27 Institutes and Centers and is a component of the U.S. Department of Health and Human Services. NIH is the primary federal agency conducting and supporting basic, clinical, and translational medical research, and is investigating the causes, treatments, and cures for both common and rare diseases. For more information about NIH and its programs, visit www.nih.gov.
NIH…Turning Discovery Into Health®






Post Comment